 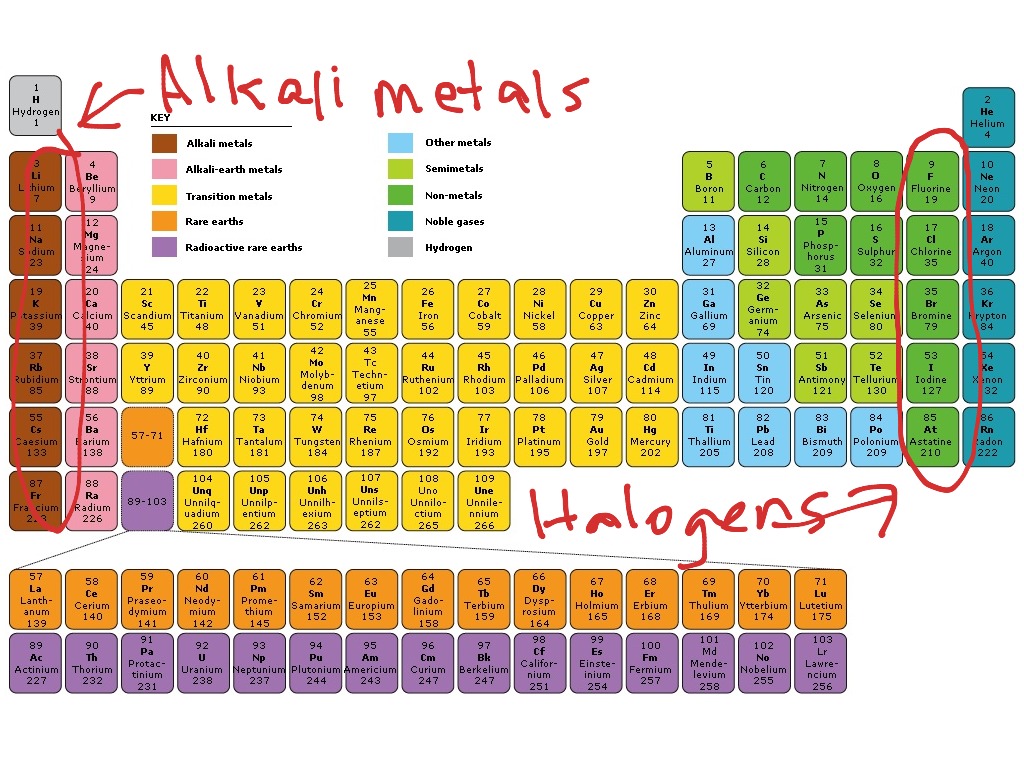
The same goes for bromine (4s 24p 5), iodine (4s 24p 5), and astatine (5s 25p 5).Īll members of the halogen family have the same valence-shell electron configurations, and thus tend to bond in much the same way. Note that only the energy level changes, but not the electron configuration at the highest energy level. Fluorine, on row 2, has a valence-shell configuration of 2s 22p 5 while that of chlorine is 3s 23p 5. In any case, electron configuration is one of the ways halogens can be defined: all have seven valence electrons.īecause the rows in the periodic table indicate increasing energy levels, energy levels rise as one moves up the list of halogens. These are often referred to as the "outer shell" of an atom, though the actual structure is much more complex. The valence electrons, which occupy the highest energy levels of an atom, are the electrons that bond one element to another. In the eight-group system, group number designates the number of valence electrons. In many ways, this is less useful than the system of 18 group numbers however, it does have one advantage. The remaining eight columns are the only ones assigned group numbers. The 40 transition metals in the center are not designated by group number, nor are the lanthanides and actinides, which are set apart at the bottom of the periodic table. In many American versions of the chart, there are only eight groups, sometimes designated with Roman numerals. Many chemists outside the United States refer to these as 18 different groups of elements however, within the United States, a somewhat different system is used. There are always 18 columns however, the way in which these are labeled differs somewhat from place to place. On the periodic table, as displayed in chemistry labs around the world, the number of columns and rows does not vary, since these configurations are the result of specific and interrelated properties among the elements. They are listed below, along with chemical symbol and atomic number: HOW IT WORKS The Halogens on the Periodic TableĪs noted, the halogens form Group 7 of the periodic table of elements. The applications of halogens are many and varied, including some that are dangerous, controversial, and deadly. The first four of these are widely used, often in combination the last, on the other hand, is a highly radioactive and extremely rare substance. Known collectively by a term derived from a Greek word meaning "salt-producing," the halogen family consists of five elements: fluorine, chlorine, bromine, iodine, and astatine. 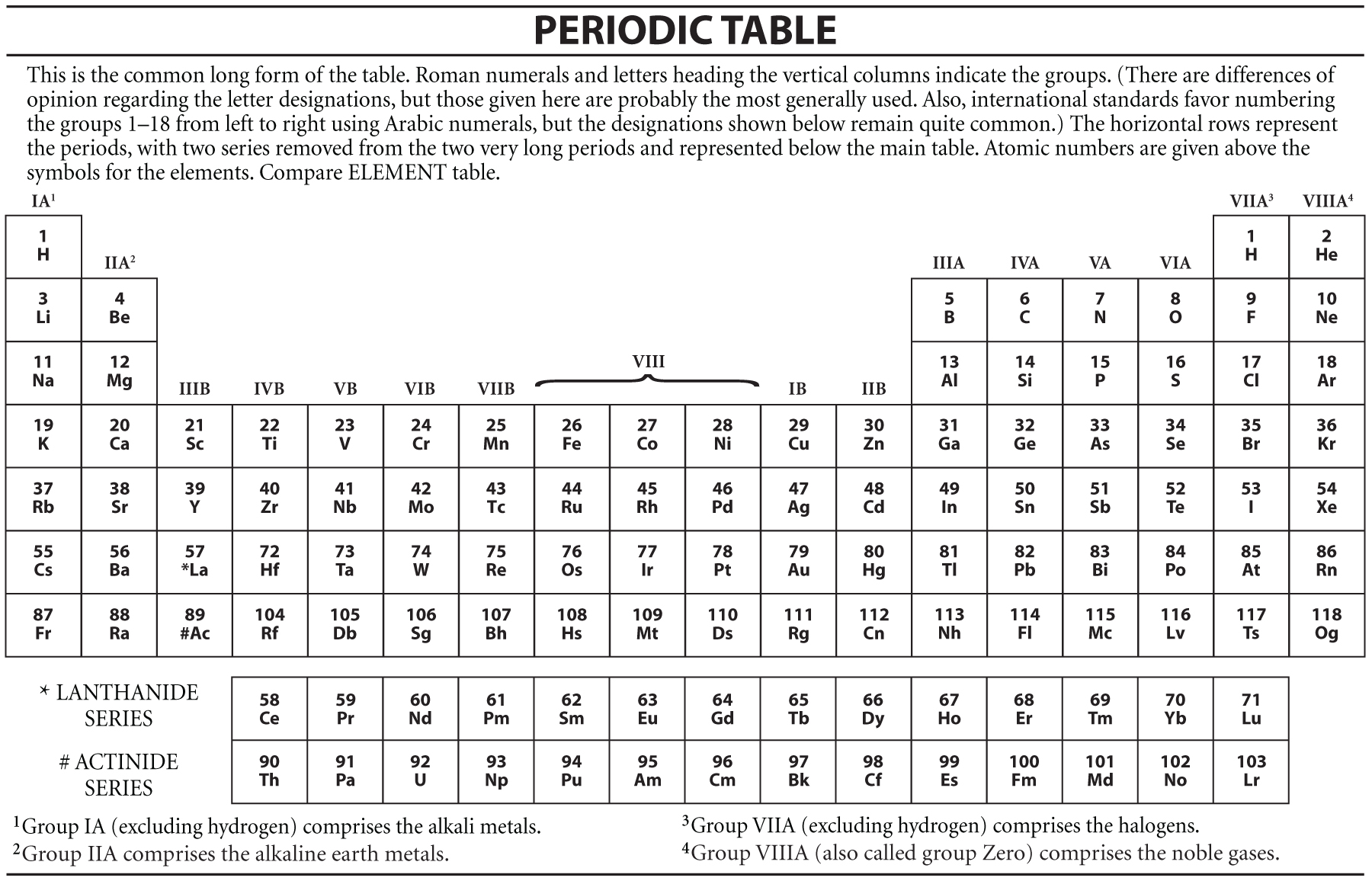
Table salt, bleach, fluoride in toothpaste, chlorine in swimming pools -what do all of these have in common? Add halogen lamps to the list, and the answer becomes more clear: all involve one or more of the halogens, which form Group 7 of the periodic table of elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
